
Our HYDAK® hydrophilic coating technology offers industry-leading performance in lubricity, durability, and particulates.

Only a few microns thick and easily mold to any device substrate.

Bi-laminar coating platform used to covalently bond the coating to the substrate material.

Methods include: ethylene oxide (EtO), gamma and e-beam.

Coatings apply to nearly all polymerics, metals, thin films & silicone.
 |
 |
|
| Ultraviolet (UV) Lighting System | Type of Curing Machine | Heat via Oven |
| Neurovascular, Cardiovascular, Peripheral Vascular | Market Applications | Neurovascular, Cardiovascular, Peripheral Vascular, Ophthalmology |
| Yes | Bi-laminar Platform | Yes |
| Ethylene Oxide (EtO), E-Beam and Gamma | Sterilization Methods | Ethylene Oxide (EtO), E-Beam and Gamma |
| Outer Diameter (OD) | Inner Diameter (ID) vs Outer Diameter (OD) Coating Application | Inner Diameter (ID) & Outer Diameter (OD) |

 Medical Device Executive and Serial Entrepreneur
Medical Device Executive and Serial Entrepreneur 




Our coatings can be applied to almost all polymerics. If you do not see your substrate listed below, please reach out.


Biocoat HYDAK technology allows for the coating of hydrophilic films used in medical devices. This capability allows for the processing of:

To support the development of cutting-edge medical devices, Biocoat has added dedicated in-house plasma capabilities. The use of plasma treatment supports the coating process by:
By definition, hydrophilic coatings are lubricious and will reduce the amount of friction that is required to maneuver a medical device through the body. Internal research at Biocoat has shown that devices coated with our hydrophilic coatings will routinely reduce the friction force by approximately 95% when compared to an uncoated device.
Due to the many twists and turns during insertion of a device, the coating needs to maintain the same level of performance throughout the entire procedure. This ensures that the coating will remain effective for the duration of the device’s use and still allow for a smooth recovery of the device once the procedure is complete.
Particulates are a relatively new measure of coating performance that is being driven by various regulatory agencies to understand what types of foreign materials are being introduced to the body and what their long-term impacts are to the patient.
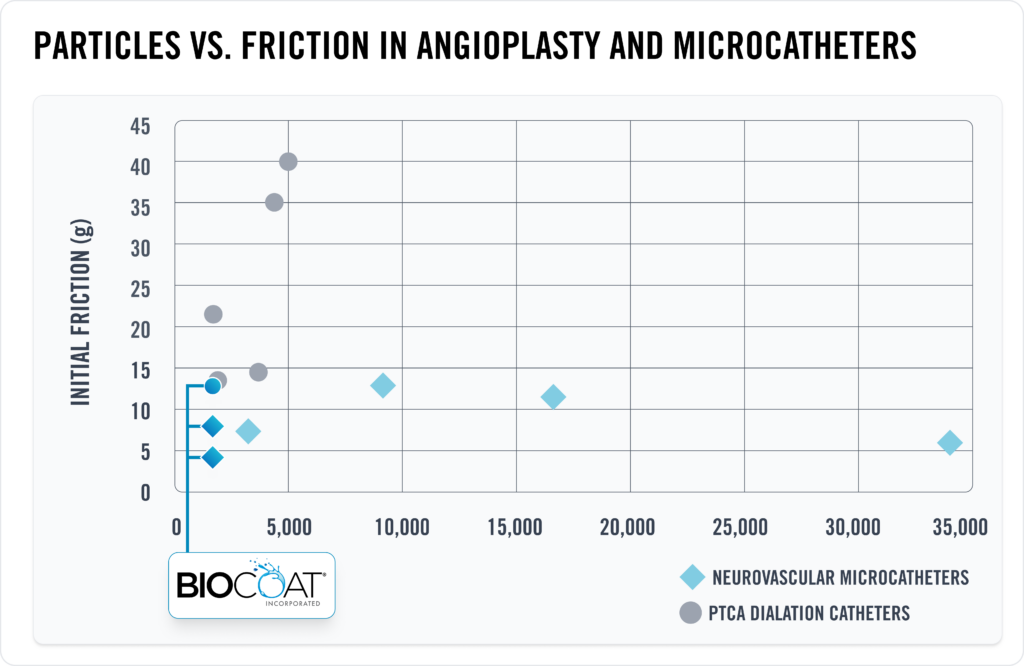
Biocoat R&D team tested 12 commercially available catheters (6 neurovascular microcatheters and 6 PTCA dilatation catheters). Three of the catheters were coated with Biocoat HYDAK® coatings, which tested as some of the best-in-class materials for both low particulates and low initial friction results in this study.
– Data on file.
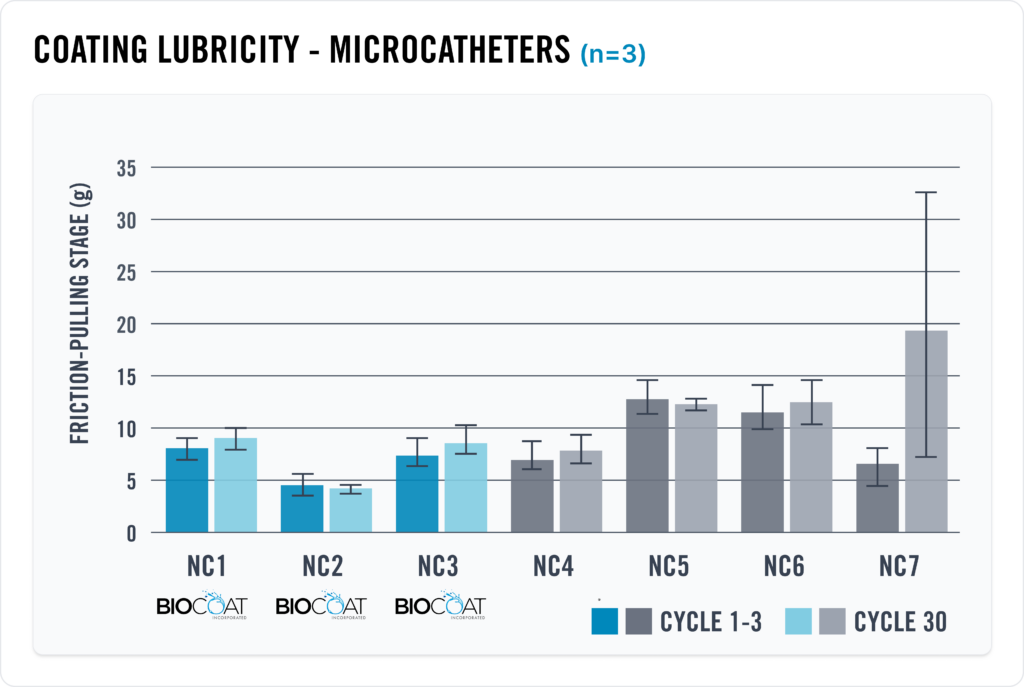
– In this chart, the Biocoat R&D team purchased seven (7) different commercially available neurovascular microcatheters, two (2) of which were coated with Biocoat HYDAK® technology. The Biocoat team measured both the lubricity and durability of each of the microcatheters in an effort to determine where HYDAK® stands in relation to industry competitors. Our testing proves that HYDAK® exhibits best-in-class lubricity and durability results.
– The chart also shows the performance of Biocoat HYDAK UV coating performance in comparison to the seven (7) commercially available microcatheters.
– Data on file.
The latest news, articles, and resources, sent to your inbox.

Biocoat has extensive experience in applying HYDAK® hydrophilic coatings to nearly all polymeric and metallic substrates, including, but not limited to, the items listed below.
Devices coated with HYDAK can be sterilized using several methods, including ethylene oxide (EtO), e-beam, and gamma. Additionally, our projects are coated in class seven clean rooms that meet all GMP standards.
HYDAK’s high-performance capabilities are a result of using a bi-laminar coating platform, which is used to covalently bond the coating to the substrate material. HYDAK’s basecoat is hydrophobic and is used to “normalize” the substrate material and to prepare the surface for a chemical bonding to the lubricious topcoat. Our topcoat provides a lubricious “slippery” surface on the medical device, which allows the device to easily move through the body. This cross-linked chemistry enables HYDAK to remain durable and lubricious throughout the entire procedure.
HYDAK adds only a few microns to the overall diameter of the device surface, which allows for our coatings to be flexible as the device travels through the body’s tortuous paths.
There are three main criteria that are used to evaluate the performance of a hydrophilic coating: friction, durability and particulates. Our HYDAK® Coating Technology platform outperforms other chemistries and delivers superior lubricity with extremely low particulate counts.
Friction
By definition, hydrophilic coatings are lubricious and will reduce the amount of friction that is required to maneuver a medical device through the body. Internal research at Biocoat has shown that devices coated with our hydrophilic coatings will routinely reduce the friction force by approximately 95% when compared to an uncoated device.
Durability
Due to the many twists and turns during insertion of a device, the coating needs to maintain the same level of performance throughout the entire procedure. This ensures that the coating will remain effective for the duration of the device’s use and still allow for a smooth recovery of the device once the procedure is complete.
Particulates
Particulates are a relatively new measure of coating performance that is being driven by various regulatory agencies to understand what types of foreign materials are being introduced to the body and what their long-term impacts are to the patient.
Biocoat hydrophilic coatings are custom engineered to meet your design parameters. As the only hydrophilic coating provider to offer industry-leading performance using both UV and Thermal heat curing systems, we can test to find the solution that maximizes the effectiveness of your device. Or we can work with your existing coating systems to support your next-generation product.